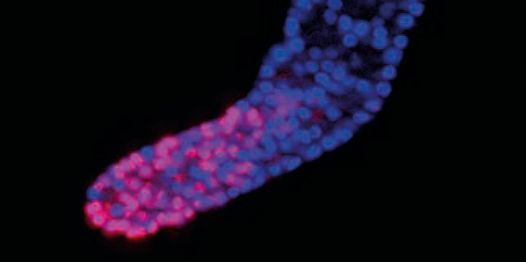
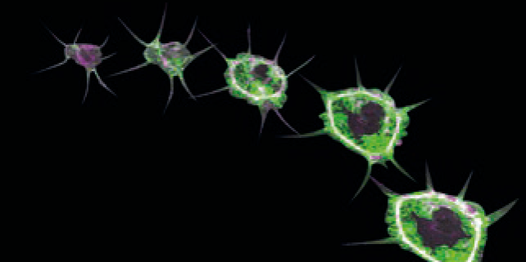
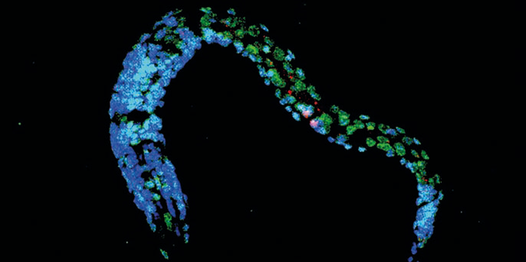
During the aging process, different stress response pathways are tightly coordinated, thereby ensuring physiological integrity especially upon metabolic changes.
Recent studies have identified cell nonautonomous regulation of cellular stress responses, suggesting interorgan communication mechanisms that are intricately balanced for integrity and maintenance of the entire organism.
Scientists of Research Area 2 aim to unravel the interplay between homeostasis pathways at the molecular, cellular, and organismal level, as well as to define adaptation mechanisms in response to aging or inherited, disease-associated mutations.
These include the findings of the Partridge, Leptin and Eming groups on the roles of TOR complexes in epidermal morphogenesis and regeneration in flies, mice and humans (Kakanj et al., Nat Protoc. 2020). The Eming group dissected how the TORC2 complex controls the skin barrier function (Ding et al., JACI. 2020) and, together with the Wickstrom group, identified TORC2 as critical regulator of metabolism and longevity of hair follicle stems cells (Kim et al., Cell Metab. 2020). Metabolism is strongly influenced by food intake, which requires adjustments of the cellular proteostasis machineries. The Hoppe group uncovered an unexpected microRNA-mediated mechanism through which food perception impacts the organism’s proteostasis (Finger et al., Nat Metab. 2020). In mammals, astrocytes respond to alterations in the nutritional intake. The Bruning group uncovered that this glial cell type regulates an adaptive stress response that improves systemic glucose homeostasis and memory formation (Timper et al., Cell Metab. 2020). The memory effect of nutrition intake can actually trigger lifelong effects on longevity and metabolic remodeling as demonstrated by the Partridge group (Hahn et al., Nat Metab. 2019). Environmental factors such as UV or mechanical stress can lead to genome damage. Collaborating with the Niessen team, the Wickstrom group demonstrated how the chromatin protects itself through epigenetic mechanisms from the impact of mechanical stress (Nava et al., Cell. 2020). Epigentic regulators are not only important for modifying the consequence of environmental stress on the genome but were shown by the Schumacher group to also mediate the recovery of protein biosynthesis and homeostasis that is required for animals to survive the consequences of DNA damage (Wang et al., Nat Struct Mol Biol. 2020). The Uhlirova group showed further that response mechanisms to oxidative stress regulate the renewal capacities of intestinal stem cells that are essential for maintaining the organism’s homeostasis (Mundorf et al., Cell Rep. 2019). Stem cells of the germline are also impacted by niche signaling as the Schumacher group revealed in the p53-mediated DNA damage response (Ou et al., Dev Cell. 2019). They further showed that stress responses in the intestine regulate the genome quality control in the germline consequently impacting aneuploidy in the offspring (Soltanmohammadi et al. Nat Comms, 2022). The Vilchez group showed that germ stem cells respond to reduced temperature and then impact the lifespan of animals (Ju Lee et al., Nat Metab. 2019). On the organ level, the Schermer and Benzing groups identified a new molecular mechanism that links aging-dependent morphological changes to chronic kidney disease (Butt et al. Nat Metab. 2020). In addition, the Frezza group could show the acute response of a metabolic dysregulation in the kidney in the progression of kidney cancer (Zecchini et al., Nature 2023).


Prof. Dr. Christian Frezza
Principal Investigator
Alexander von Humboldt Professor of Metabolomics in Aging
Head of Research Area 2
christian.frezza[at]uni-koeln.de
Metabolomics in aging
Room: 4.068
CECAD Research Center
Joseph-Stelzmann-Str. 26
50931 Köln

Dr. Anne Schaefer
Principal Investigator
Director of MPI for Biology of Ageing
Head of Research Area 2